Handling OOS, OOT & Deviations in Real Labs: Expert Strategies Unveiled
Are you struggling with unexpected Out of Specification (OOS), Out of Trend (OOT), or deviations in your real lab results? You’re not alone.
These issues can disrupt your workflow, cause delays, and even risk compliance. But what if you could handle them smoothly and confidently every time? You’ll discover practical strategies to manage OOS, OOT, and deviations effectively. By understanding the root causes and applying proven techniques, you’ll regain control over your lab processes and ensure reliable outcomes.
Keep reading to transform challenges into opportunities for improvement in your lab work.
Credit: gmpinsiders.com
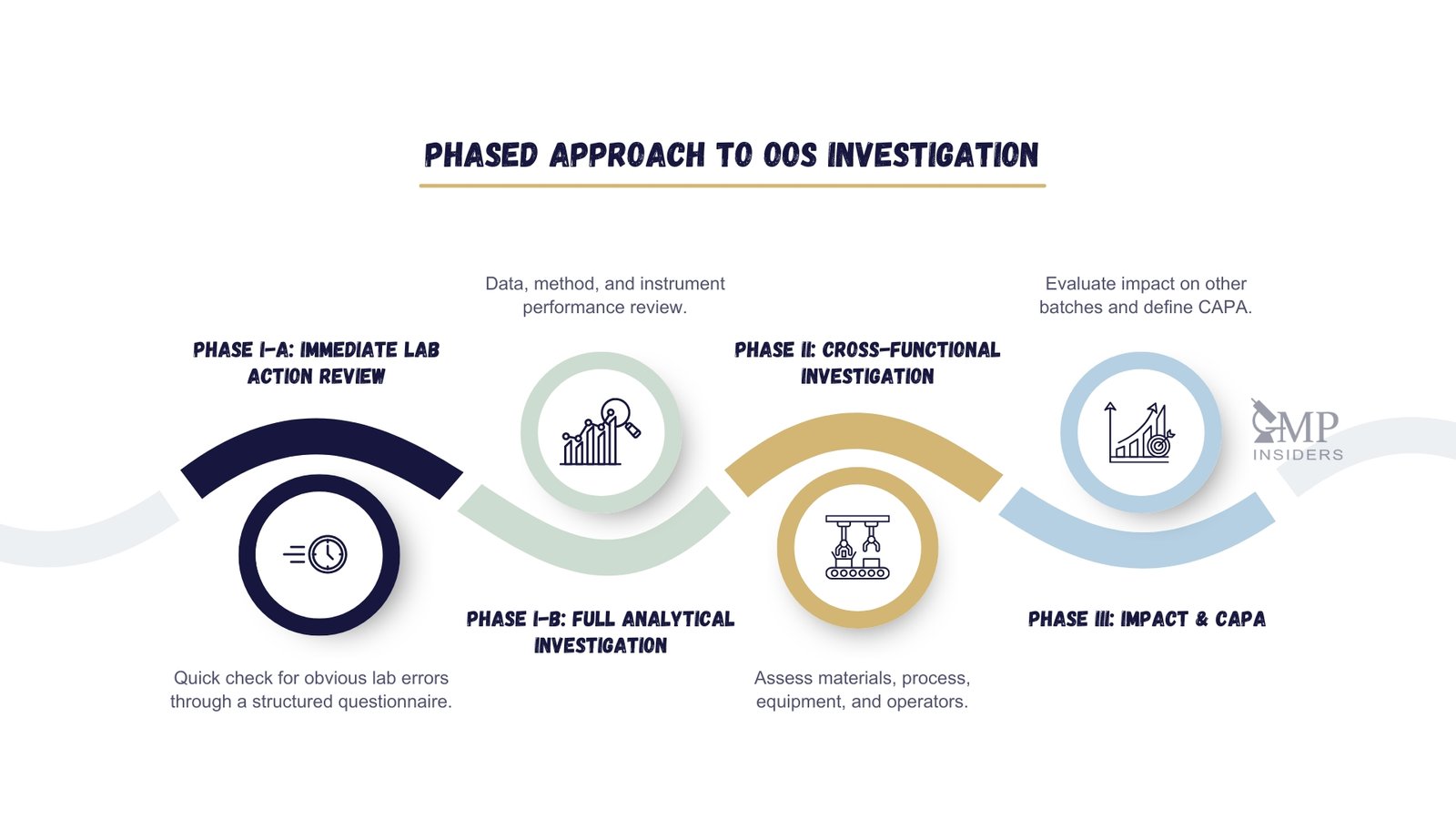
Common Causes Of OOS, OOT, And Deviations
Out-of-Specification (OOS), Out-of-Trend (OOT), and deviations are common challenges in real lab settings. These issues can affect the accuracy and reliability of test results. Identifying the root causes helps maintain high-quality standards. Labs must carefully track and analyse these causes to reduce errors and improve processes.
Instrument And Equipment Issues
Faulty instruments often lead to OOS and OOT results. Calibration errors cause inaccurate measurements. Equipment wear and tear reduces precision over time. Poor maintenance can result in malfunctioning devices. Using outdated tools also affects data reliability.
In this case, calibration and validation must be performed perfectly at regular intervals.
Sample Handling Errors
Incorrect sample collection causes deviations in results. Contamination during handling alters sample integrity. Wrong labelling leads to mix-ups and false data. Improper storage conditions change sample properties. Delays in testing can degrade sample quality.
Environmental Factors
Temperature fluctuations impact chemical reactions in samples. Humidity levels may affect instrument performance. Dust or dirt in the lab causes contamination. Lighting conditions can influence visual inspections. Noise and vibrations disturb sensitive equipment.
Method And Procedure Variations
Deviations arise from inconsistent test methods. Skipping steps or rushing procedures causes errors. Using unapproved methods leads to unreliable results. Lack of staff training results in procedural mistakes. Poor documentation hampers troubleshooting efforts. Validating methods and processes in chemical analysis is essential to prevent Out-of-Specification (OOS) and Out-of-Trend (OOT) results.
Detecting And Documenting Anomalies
Detecting and documenting anomalies in real labs is crucial for reliable results and safety. Anomalies can affect test outcomes and lead to wrong decisions. Early detection helps address problems before they grow. Proper documentation creates a clear record for review and action. Labs must have clear steps to quickly and accurately identify and log any deviations.
To detect errors and document the perfect process, it is essential to understand accuracy and precision in chemical analysis to avoid further mistakes.
Early Warning Signs
Look for unusual patterns in data or equipment behaviour. Sudden changes in temperature, pressure, or timing can signal problems. Listen for odd noises or observe visual defects. Staff should report any irregularities immediately. Early signs often appear before the issue becomes serious. Training teams to recognise these helps stop errors fast.
Accurate Record Keeping
Keep detailed notes of every anomaly detected. Record the date, time, and conditions of the event. Describe what was unusual and who noticed it. Include any steps taken to fix the problem. Clear, organised records support analysis and future prevention. They also meet compliance and audit needs.
Role Of Laboratory Information Management Systems
Laboratory Information Management Systems (LIMS) streamline anomaly tracking. LIMS stores data securely and makes retrieval easy. It can alert staff to deviations in real time. Automated reports save time and reduce errors. LIMS supports consistent documentation and helps maintain quality standards. It is a vital tool for modern labs.
Root Cause Analysis Techniques
Root cause analysis techniques help find the main reasons for Out of Specification (OOS), Out of Trend (OOT), and deviations in real labs. These methods guide teams to understand problems deeply. Fixing the root cause stops the problem from happening again. Simple tools can make complex issues clearer.
Fishbone Diagrams
Fishbone diagrams show the causes of a problem in a visual way. They look like a fish skeleton. The main problem is the head. Branches are categories of causes, such as people, methods, machines, materials, and environment. Teams brainstorm under each branch. This helps find all possible causes quickly. It organises thoughts and reveals hidden issues.
5 Whys Method
The 5 Whys method digs deeper by asking "Why?" five times. Each answer leads to the next question. This simple technique uncovers the root cause step by step. It avoids blaming people and focuses on process problems. Teams can use this method alone or with other tools. It saves time and clarifies the real issue.
Fault Tree Analysis
Fault Tree Analysis (FTA) uses a top-down approach. It starts with the main failure at the top. It breaks down into smaller causes below. FTA uses logic gates like AND and OR to connect causes. This method shows how different factors combine to cause a failure. It is useful for complex or safety-critical problems. Clear visuals help teams understand and solve issues.
Corrective And Preventive Actions
Corrective and Preventive Actions (CAPAs) are vital in managing Out of Specification (OOS), Out of Trend (OOT), and deviations in real labs. They help identify problems and stop them from happening again. CAPAs ensure lab results stay reliable and quality standards are met. Labs must act quickly and carefully to fix issues and prevent future errors.
Developing Effective CAPAs
Start by finding the root cause of the problem. Use data and team input to understand what went wrong. Define clear actions that fix the issue and prevent its return. Set simple, measurable goals for each action. Avoid vague or complex steps. Make sure the plan covers both correction and prevention.
Implementation Strategies
Assign tasks to the right team members. Provide training or resources if needed. Keep the timeline clear and realistic. Document every step for tracking and review. Communicate regularly with all involved to ensure smooth execution. Adjust the plan if new information arises.
Monitoring And Follow-up
Track the results of CAPA actions carefully. Use audits or tests to check effectiveness. Look for signs that the problem is solved. If issues persist, review and revise the plan. Keep records of monitoring activities. Regular follow-up helps maintain lab quality and compliance.
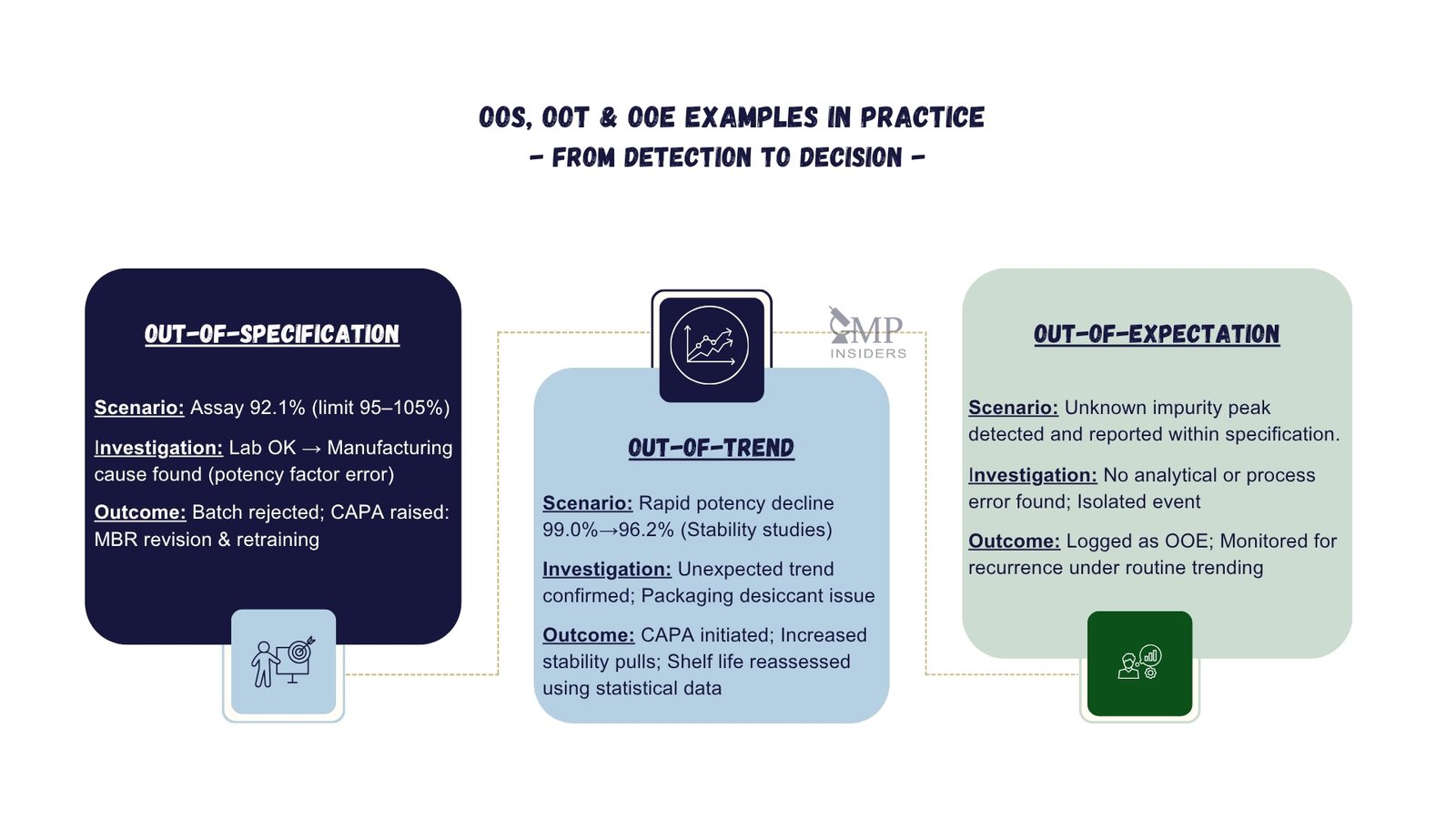
Training And Team Collaboration
Handling Out of Specification (OOS), Out of Trend (OOT), and deviations in real labs requires strong training and team collaboration. Proper training equips lab staff to recognise and manage these issues quickly. Teamwork ensures smooth communication and faster resolution.
Training and collaboration create a proactive environment. Staff learn to prevent errors and handle problems calmly. This approach reduces downtime and improves lab accuracy.
Skill Development Programs
Skill development programs train lab staff on testing procedures and quality standards. Regular workshops teach how to spot OOS, OOT, and deviations early. Hands-on practice builds confidence in handling unexpected results. These programs keep skills fresh and updated with industry rules.
Communication Best Practices
Clear communication is key to managing lab issues. Teams should share information openly and promptly. Daily briefings and digital logs help track deviations. Using simple language avoids confusion. Good communication speeds up problem-solving and prevents mistakes.
Fostering A Quality Culture
Building a quality culture means everyone values accuracy and safety. Leaders set examples by following procedures strictly. Staff feel responsible for reporting problems without fear. Regular feedback and recognition encourage care and attention. A strong quality culture lowers the risk of errors and improves lab results.
Credit: gmpinsiders.com
Leveraging Technology For Better Control
Handling out-of-specification (OOS), out-of-trend (OOT), and deviations in real labs requires precise control. Technology offers tools that improve accuracy and speed. Labs can reduce errors and increase efficiency by using modern systems. These tools help detect problems early and maintain high-quality standards.
Automation And Real-time Monitoring
Automation reduces manual work and limits human error. Machines perform repetitive tasks with consistency. Real-time monitoring tracks lab processes continuously. This instant data helps staff respond quickly to issues. Automation keeps workflows smooth and reduces delays.
Data Analytics And Trending
Data analytics identifies patterns in lab results over time. Trending highlights deviations and outliers early. This insight helps predict potential problems. Labs can act before errors affect products. Analytics improves decision-making with clear, visual reports.
Digital Documentation Tools
Digital tools replace paper records and improve accuracy. They make storing and retrieving data faster. Electronic documentation ensures traceability for audits and reviews. These tools reduce the risk of lost or altered records. Digital records streamline lab compliance and reporting.
Regulatory Expectations And Compliance
Regulatory expectations play a key role in managing Out of Specification (OOS), Out of Trend (OOT), and deviations in real labs. Following these rules ensures product safety and quality. Compliance helps avoid penalties and builds trust with regulators. Labs must be ready to prove their processes meet standards at all times.
Guidelines From Regulatory Bodies
Regulatory bodies like the FDA and EMA set clear rules for handling OOS, OOT, and deviations. They require thorough investigations and proper documentation. Labs must identify root causes quickly and take corrective actions. Accurate records and transparency are crucial. Regular updates to procedures keep labs aligned with current expectations.
Audit Preparation Tips
Prepare for audits by keeping all documents organised and easy to find. Train staff on procedures and compliance requirements. Review previous OOS and deviation reports for completeness. Conduct internal audits to spot gaps early. Clear records and confident staff create a positive audit impression.
Handling Regulatory Inspections
During inspections, stay calm and answer questions honestly. Provide requested documents promptly. Show how your lab follows guidelines for OOS, OOT, and deviations. Demonstrate corrective actions taken and improvements made. Cooperation and transparency build confidence with inspectors and support compliance.
Frequently Asked Questions
What Are OOS, OOT, And Deviations In Real Labs?
OOS means Out of Specification, OOT means Out of Trend, and deviations refer to any unplanned changes. These terms indicate issues in lab results or processes that need investigation to maintain quality and compliance.
How To Handle OOS Results Effectively In Laboratories?
Investigate all possible causes, document findings, and follow SOPs strictly. Perform retesting if necessary and report the outcome to ensure product safety and regulatory compliance.
Why Is Managing Oot Important In Lab Testing?
OOT results signal unusual trends that could indicate equipment or process issues. Early detection helps prevent product failures and maintains data integrity.
What Steps Should Be Taken For Deviations In Lab Processes?
Identify the deviation cause promptly, document it, and assess its impact. Implement corrective actions and prevent recurrence to ensure consistent lab operations.
Conclusion
Handling OOS, OOT, and deviations takes careful attention in real labs. Clear steps help spot issues fast and fix them right. Teams should keep good records and follow set rules. This keeps lab work safe and results trustworthy. Practice makes it easier to manage problems well.
Staying alert and ready helps labs run smoothly every day.


