Regulatory Writing & Dossier Basics: Essential Tips for Success
Are you stepping into the world of regulatory writing and feeling overwhelmed by all the rules and documents? You’re not alone.
Understanding the basics of regulatory writing and dossiers is key to making your work smoother and more effective. You’ll discover clear, simple explanations that break down complex ideas into easy steps. By the end, you’ll feel more confident handling regulatory documents and know exactly what makes a strong dossier.
Ready to take control of your regulatory writing skills? Let’s dive in.
Credit: www.freyrsolutions.com
Role Of Regulatory Writing
Regulatory writing plays a crucial role in the drug development process. It involves creating clear, accurate documents that explain a drug’s safety and effectiveness. These documents help regulatory agencies decide if a drug can be approved. Writers must follow strict guidelines to ensure the information is easy to understand and complete.
Good regulatory writing helps speed up drug approval. It reduces errors and questions from regulators. Clear writing builds trust and shows that the drug is well-studied. It supports the work of scientists, doctors, and companies involved in making the drug.
Purpose of Drug Approval
The main purpose of regulatory writing is to support drug approval. It presents all the data from clinical trials and research. This data shows how the drug works and if it is safe for patients. The documents must explain the science clearly.
Regulatory writing helps agencies review the drug quickly. It ensures that the drug meets legal and safety standards. Without clear documents, approval can be delayed or denied.
Key Documents And Formats
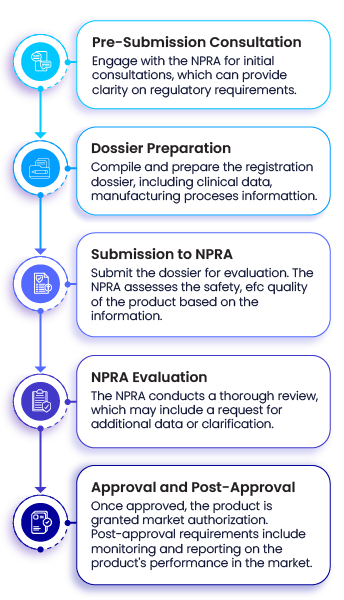
Several key documents form the core of regulatory writing. The Common Technical Document (CTD) is the standard format used worldwide. It organizes information into modules covering quality, safety, and efficacy.
Other important documents include clinical study reports, investigator brochures, and risk management plans. Each document follows specific templates and guidelines. This uniformity helps regulators find information easily and compare data across drugs.
Components Of A Dossier
A dossier is a detailed collection of documents. It provides all necessary information about a product for regulatory review. Understanding its components helps in preparing clear and complete submissions.
Each part of the dossier serves a specific purpose. It ensures that the product meets safety, quality, and efficacy standards. Regulatory writers play a key role in organizing this information.
Module Structure
The dossier follows a structured format divided into modules. Each module contains related documents and data. This format makes it easier for regulators to review the submission.
There are usually five main modules. They cover administrative information, quality data, non-clinical study reports, clinical study reports, and regional information. This organization helps keep the dossier clear and logical.
Common Content Requirements
Each dossier must include specific content to meet regulatory standards. Basic details include product description, manufacturing process, and labeling.
Safety data, such as toxicology and clinical trial results, are essential. Quality information includes raw materials, packaging, and stability studies. Accurate and complete data reduces review time and increases approval chances.
Writing Style And Clarity
Writing style and clarity form the foundation of regulatory documents and dossiers. Clear writing helps readers understand complex information quickly. It reduces errors and speeds up review processes.
Regulatory writing demands simplicity and accuracy. Writers must express ideas in a straightforward way. This makes the documents accessible to people from different backgrounds.
Conciseness And Precision
Concise writing uses fewer words without losing meaning. It avoids unnecessary details that confuse readers. Precision means using exact terms and figures. This approach ensures information is clear and direct.
Every sentence should serve a purpose. Short sentences make content easier to read. Avoid repeating ideas or adding filler words. Focus on delivering facts clearly and briefly.
Avoiding Ambiguity
Ambiguity causes misunderstanding and delays in decision-making. Use specific words instead of vague terms. Define technical terms or abbreviations clearly. This helps avoid confusion and misinterpretation.
Review sentences to remove double meanings. Use active voice to make statements clear. Ambiguity can lead to costly mistakes in regulatory submissions.
Compliance With Guidelines
Compliance with guidelines is essential in regulatory writing and dossier preparation. It ensures documents meet legal and scientific standards. Proper compliance helps avoid delays and rejections in submissions. It also builds trust with regulatory authorities. Understanding and following these guidelines is a key part of successful regulatory processes.
Ich And Regional Standards
The International Council for Harmonisation (ICH) sets global standards for drug development. These guidelines help align regulatory requirements across countries. Each region may have specific rules beyond ICH standards. For example, the FDA in the USA and EMA in Europe have their own guidelines. Knowing both ICH and regional standards ensures dossiers meet all necessary criteria. This knowledge reduces errors and speeds up approval times.
Adhering To Submission Rules
Submission rules define how documents must be formatted and presented. These rules cover file types, document structure, and required content. Following these rules avoids administrative issues during review. Each authority has strict submission protocols. Meeting these requirements shows professionalism and attention to detail. Proper adherence also helps reviewers find information quickly. This leads to smoother evaluation and faster decisions.
Effective Document Management
Effective document management is key in regulatory writing and dossier preparation. It keeps all documents organized and easy to find. This process saves time and reduces errors. Good management helps teams work smoothly and meet deadlines.
Understanding how to control versions and collaborate well improves document quality. It also ensures compliance with regulatory standards. Clear methods for managing documents are essential in this field.
Version Control
Version control tracks all changes made to a document. It shows who edited the file and when. This avoids confusion from multiple versions floating around. Using version control tools keeps documents up-to-date and accurate.
Each version is saved separately. Teams can revert to older versions if needed. This system protects against data loss and errors. It also helps reviewers see the document's progress over time.
Collaboration Techniques
Good collaboration means clear communication and shared access. Teams use platforms that allow multiple users to work on documents together. This speeds up the review and approval process.
Assigning roles and responsibilities avoids overlap and mistakes. Regular meetings and updates keep everyone informed. Effective collaboration ensures the dossier is complete and meets all requirements.
Common Challenges And Solutions
Regulatory writing and dossier preparation involve many challenges. These challenges can slow down the approval process. Understanding common issues helps create better solutions. It also improves the quality of submissions.
Clear communication and accurate data are vital. Sometimes, missing information or reviewer questions cause delays. Addressing these problems quickly keeps projects on track. Below are key challenges and practical ways to handle them.
Handling Data Gaps
Data gaps occur when important information is missing. This can happen due to incomplete studies or lost records. Such gaps create confusion for reviewers. They may doubt the reliability of the dossier.
Start by identifying all missing data early. Communicate clearly with the team to fill gaps fast. Use alternative data sources if possible. Explain any limitations honestly in the submission. This builds trust and avoids future questions.
Addressing Reviewer Comments
Reviewers often request clarifications or additional details. Their comments can seem strict or complex. Ignoring or delaying responses causes further delays. Respond to each comment carefully and respectfully.
Organize comments by priority and topic. Assign clear responsibilities to team members. Provide clear, concise answers with supporting evidence. Keep the tone professional and polite. This approach speeds up approval and builds a good reputation.
Tips For Successful Submissions
Successful regulatory submissions demand clear planning and strict quality control. These tips help avoid common mistakes and delays. Follow a simple, organized approach for better outcomes.
Early Planning Strategies
Start preparing the dossier early. Gather all required documents well before deadlines. Understand the regulatory guidelines specific to your product. Create a detailed timeline with milestones for each stage. Assign clear roles and responsibilities to your team. Early planning reduces last-minute stress and errors.
Quality Assurance Practices
Review every document carefully for accuracy and completeness. Use checklists to ensure all sections meet regulatory standards. Perform internal audits before submission to catch errors. Maintain version control to track changes effectively. Consistent quality checks build confidence with regulators and speed up approvals.
How Chemistry Job Insight Can Help You with Regulatory Writing & Dossier Basics
Enhancing Your Skills in Regulatory Writing and Dossier Preparation
Mastering regulatory writing and dossier basics involves more than understanding the role of regulatory writing or the components of a dossier; it requires hands-on practice with writing style, clarity, and compliance with guidelines. Practical learning opportunities, such as workshops or online courses, can provide invaluable experience in organizing complex information while maintaining precision and reliability—qualities highly valued in this field.
For chemists keen on advancing their expertise, focusing on effective document management techniques can streamline the submission process and reduce common challenges. Engaging with communities or resources like Chemistry Job Insight offers actionable insights and practical knowledge to navigate these areas confidently.
- Participate in case study reviews to apply compliance requirements in real scenarios.
- Practice drafting sections of dossiers to enhance clarity and adherence to guidelines.
- Utilize document management tools to improve organization and version control.
For further guidance or clarification, reaching out via contact@chemistryjobinsight.com can connect you with expert advice tailored to regulatory writing and dossier preparation challenges.
Frequently Asked Questions
What Is Regulatory Writing In Pharmaceutical Industry?
Regulatory writing involves creating clear, precise documents for drug approval. It ensures compliance with regulatory standards. Writers prepare dossiers, clinical study reports, and safety documents to support product registration worldwide.
Why Are Dossiers Important In Drug Approval Process?
Dossiers compile all scientific and administrative data for regulatory review. They prove a drug’s safety, efficacy, and quality. Accurate dossiers speed up the approval process and ensure regulatory compliance.
What Documents Are Included In A Regulatory Dossier?
A regulatory dossier includes clinical study reports, product information, manufacturing details, and safety data. These documents support a drug’s approval by regulatory authorities. Each part must be precise and well-organized.
How Does Regulatory Writing Impact Drug Development?
Regulatory writing ensures all documentation meets legal standards. It facilitates communication between sponsors and regulatory agencies. Proper writing reduces delays and helps drugs reach the market faster.
Conclusion
Regulatory writing and dossier preparation form the backbone of successful submissions. Clear documents help reviewers understand data quickly. Following guidelines ensures compliance and speeds approval. Strong writing reduces errors and saves time. Everyone involved benefits from well-organized dossiers. Keep learning and practicing to improve your skills.
The process may seem complex, but steady effort pays off. Stay focused, be precise, and maintain clarity throughout. This approach leads to better communication and smoother regulatory journeys.
